Organization of the Contractile Apparatus and Costameres
Costameres
Costameres are structures at the plasma membrane, or “sarcolemma”, of striated muscle that form as myofibrils develop nearby, and that remain linked to the underlying contractile apparatus through regular connections established by cytoskeletal filaments that run from the ends and the middle of each superficial sarcomere to the membrane. They therefore appear like thick or thin “ribs” around the surface of each fiber– hence their name (see figure). In some fibers, they are crisscrossed by longitudinal structures to create a pattern resembling window frames. Costameres are used to keep the sarcolemma in register with the nearby contractile apparatus, to protect the membrane against damage during contraction, and to transmit the force of contraction radially, through the sarcolemma to the extracellular matrix and nearby muscle fibers, and from there to the tendons.
We are particularly interested in costameres because they contain high concentrations of dystrophin, the protein that is missing in young boys with Duchenne Muscular Dystrophy (see below), and they are unstable when dystrophin is absent. The mechanisms responsible for maintaining costameres are therefore likely to be linked to those that maintain a stable sarcolemma, and any changes in costameres that diminish their stability are likely to destabilize the membrane, leading to muscle disease. We have repeatedly shown this to be true in mouse and human skeletal muscles. Images of costameres in healthy muscle fiber are shown below.
Intermediate Filaments
Intermediate filaments are present in the cells of all higher organisms and contribute to the mechanical stability of many tissues, including striated muscle. Our studies indicate that intermediate filaments (IFs) help link costameres to the contractile apparatus. Desmin, the major IF protein of muscle, has been studied extensively, and desminopathies linked to mutations in the gene encoding desmin have been documented in the human population. Our laboratory is the first to show that a second class of IF proteins, the keratins, are also expressed in adult striated muscle, and are associated with the sarcolemma, perhaps through dystrophin. Consistent with this, mice missing keratin 19 have unstable costameres and a mild, though distinct, skeletal myopathy – the first evidence of this protein having an essential role in any mammalian tissue.
Intermediate filaments are also involved in regulating gene expression, probably by serving as anchors for signaling proteins that sense the environment in the subcellular domains inhabited by the filaments to which they bind. We are now completing gene array studies of desmin-null and keratin-null muscles that have identified the genes that are up- or down-regulated when one or the other IF protein is missing. Remarkably, although desmin-null fibers are significantly changed morphologically and functionally, the transcription of only ~60 genes is significantly altered (most are up-regulated). By contrast, transcription of only 5 genes is altered in the keratin 19-null. We have confirmed some of these observations by qRT-PCR, immunoblotting and immunofluorescence. These results confirm the larger role desmin plays in skeletal muscle, compared to keratin 19. They also point to desmin as a regulator of the signaling cascades involved in muscle formation and repair, consistent with its expression very early in the myogenic process.
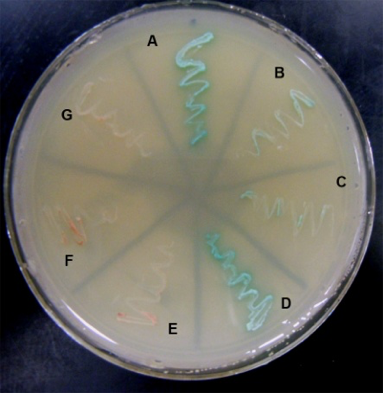
We have also been studying synemin, a large intermediate filament protein that associates with both keratin- and desmin-based filaments, and, our results show, helps to organize them in the myoplasm. In addition to its known ligands, we used the yeast two hybrid assay and direct protein-protein binding assays to show that synemin associates with the C-terminal region of titin, which is found at the middle of the sarcomere, and that this association is required for keratins to accumulate nearby. (The yeast two hybrid assay is illustrated here: blue streaks indicate specific interactions of synemin with the C-terminal domain of titin). Synemin is also present at costameres, where it binds to two costameric proteins, dystrophin and α-dystrobrevin. As synemin is an AKAP (i.e., it anchors protein kinase A), its localization in sarcomeres and costameres is likely to regulate these structures through modulating local phosphorylation. We are currently addressing this possibility by comparing normal muscle to muscle lacking synemin due to homologous recombination (synemin “knockout”), which has just been generated.
We are also using RT-PCR to identify other intermediate filament proteins of the keratin family in muscle. We hypothesize that these keratins (we have found at least 5 more, in addition to keratin 19) serve functions that complement those of keratin 19 and desmin. Our goal in studying these proteins is to learn how each contributes to the health of skeletal muscles.
Finally, we have begun to collaborate with Dr. Joseph Stains (Department of Orthopedics, University of Maryland School of Medicine) te test the roles of intermediate filament proteins in bone, which can be significant: elimination of either keratin 18 or synemin has a profound effect on reticular bone.
Microtubules
Microtubules organize along tracks of intermediate filaments and are essential for trafficking of proteins, including dystrophin and its associated glycoprotein complex, to the sarcolemma, where they concentrate in costameres. We have therefore examined the changes in the organization and composition of microtubules that occur in myofibers lacking desmin or keratin 19. Our results confirm that muscles missing one or the other of these IF proteins undergo distinct changes in tubulin modification (glu vs tyr tubulin) and in the organization of microtubules in the subsarcolemmal space, which expands when either desmin or keratin 19 is missing.
An image of the subsarcolemmal microtubules in control and keratin 19 null fibers:

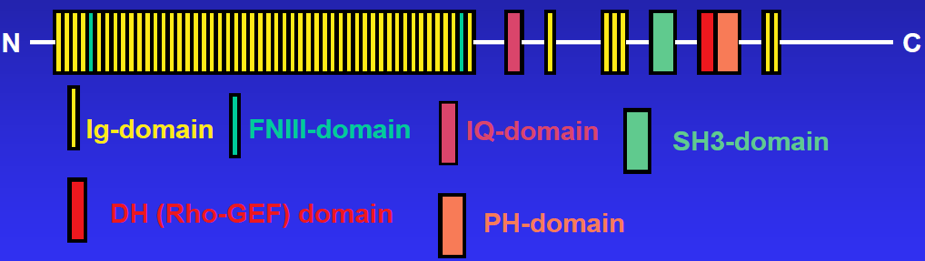
Obscurin
Obscurin is the third member of the titin superfamily to be identified in striated muscle. Like titin, a 3 MDa protein to which it is related, obscurin is a giant protein (~800 kDa) made up of many structural (mostly Ig) domains, linked to several domains involved in cellular signaling pathways (see cartoon of structure, below). Unlike titin, which is fully integrated into the contractile apparatus, obscurin surrounds the contractile structures, serving to link them to the surrounding SR (see above). We have shown that obscurin is required for the maturation of the mid-region of the sarcomere and for the efficient incorporation of the N-terminal region of titin into the sarcomere ends. We have also studied its role as a signaling protein and shown that its rhoGEF domain is capable of activating RhoA, a small GTPase. As noted above, obscurin can regulate SERCA activity in muscle through its interactions with sAnk1. Our current studies of obscurin focus on this aspect of its role in skeletal muscle.